
The ball-and-spring models of ethene/ethylene (a) and propene/propylene (b) show their respective shapes, especially bond angles. More importantly, these theoretically predicted properties have been subsequently. The Lewis structure, also called as electron dot structure, is a simplified method of representing the number of valence electrons present within an atom or a molecule. Carbon nanotubes, first discovered in the 1990s, have become one of the most eye-catching materials and have attracted great attention in the nanoscience community, because of their unique structure and truly remarkable electronic and mechanical properties. The principles involved - promotion of electrons if necessary, then hybridisation, followed by the formation of molecular orbitals - can be applied to any covalently-bound molecule.\): Ethene and Propene. The carbon monoxide is produced from the partial oxidation of carbon dioxide (CO2) or any other carbon-containing element. Each orbital holds the 2 electrons that we've previously drawn as a dot and a cross. The carbon atoms in an alkyne bond are sp hybridised, which means that they each contain two unhybridized p orbitals and two sp hybrid orbitals, according to. 
When a covalent bond is formed, the atomic orbitals (the orbitals in the individual atoms) merge to produce a new molecular orbital which contains the electron pair which creates the bond.įour molecular orbitals are formed, looking rather like the original sp 3 hybrids, but with a hydrogen nucleus embedded in each lobe. 2.1 A, and an atomic-resolution scanning transmission electron microscopy image of graphene is shown in Fig. 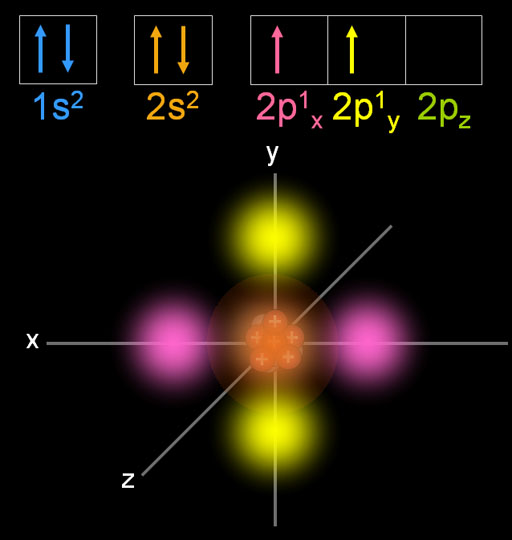
Remember that hydrogen's electron is in a 1s orbital - a spherically symmetric region of space surrounding the nucleus where there is some fixed chance (say 95%) of finding the electron. The atomic structure of graphene determined by the three bonds, which are responsible for the mechanical properties of graphene, is shown in Fig. Its first four ionisation energies, 1086.5, 2352.6, 4620.7 kJ/mol, are much higher than those of the heavier group-14 elements. For clarity, the nucleus is drawn far larger than it really is. Carbon is the sixth element, with a ground-state electron configuration of 1s 2 2s 2 2p 2, of which the four outer electrons are valence electrons. You can picture the nucleus as being at the centre of a tetrahedron (a triangularly based pyramid) with the orbitals pointing to the corners. For convenience, the energy level of 2p z is kept with no electron, though it is equivalent to the energy levels of 2p x and 2p y. Sp 3 hybrid orbitals look a bit like half a p orbital, and they arrange themselves in space so that they are as far apart as possible. You should read "sp 3" as "s p three" - not as "s p cubed". This reorganizes the electrons into four identical hybrid orbitals called sp 3 hybrids (because they are made from one s orbital and three p orbitals). In particular, the cyclo18carbon shows a great second-order hyperpolarizability and displays an obvious optical dispersion in optical nonlinearity. The electrons rearrange themselves again in a process called hybridization. Accurate calculation of molecular response properties shows that the cyclo18carbon has apparent nonlinear anisotropy due to its two-dimensional looping structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
